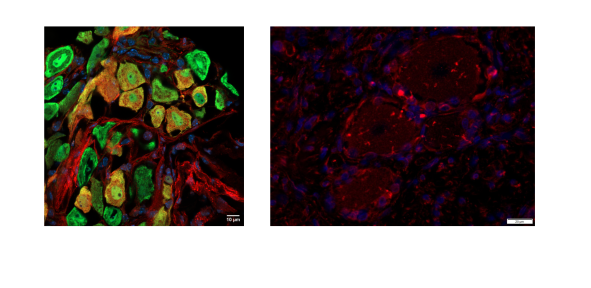
Long COVID: A study by UNamur and CHU UCL Namur unravels the biological mechanism behind the pain experienced by patients
A multidisciplinary research team from the University of Namur (UNamur) and the UCL Namur University Hospital (Godinne campus) has just published a study in the journal Acta Neuropathologica that sheds light on some of the mysteries surrounding the origin of the pain experienced by patients with long COVID. Their findings suggest that these painful symptoms may be mediated by an autoimmune response. In other words: patients produce antibodies that attack their own neurons—those responsible for pain perception and deep body sensation, located along the spine. These highly promising results represent a major scientific breakthrough that opens new avenues for better understanding the disease and, ultimately, developing a treatment targeting the painful symptoms of long COVID.